Jeringa estéril médica Lepu gana la aprobación de marca CE para uso general, vacunas COVID-19
-
2021-08-24
-
LEPU
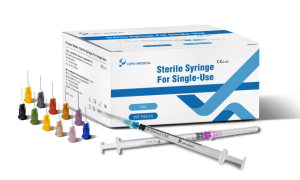
BEIJING, AUGUST 24, 2021 - Lepu Medical Technology (Beijing) Co., Ltd. (“Lepu Medical”), a leading global company specialized in developing, manufacturing and marketing high-tech medical devices and equipment, today announced that its Jeringa estéril para línea de un solo uso Ganó la aprobación de la marca CE para uso general, incluida la entrega de varias vacunas COVID-19.
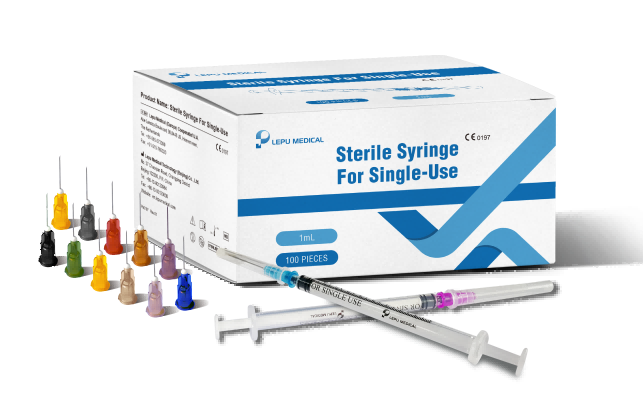
La marca CE indica que la jeringa estéril de un solo uso de Lepu Medical cumple con los estándares de seguridad y rendimiento de la categoría de la Unión Europea (UE) para su uso por profesionales de la salud para aspiración e inyección de fluidos de uso general, lo que los hace disponibles para su compra profesional en toda la UE y otros países que reconocen la marca CE.
Lepu Medical’s Jeringa estéril para línea de un solo uso has received CE Mark approval at a time of high-demand for needles suitable to vaccinate populations against the COVID-19 pandemic. Lepu Medical’s syringe series offers a variety of single-use needles for many applications, including various COVID-19 vaccines. The line includes both Luer Lock and Lock Slip syringes, a clear barrel, plunger, plunger stopper, needle, and cap. Lepu Medical’s syringe series offers a higher resistance to breakage in single-use, and latex-free pistons. The total graduated capacity of all models of syringes is 1mL, with a range of needle sizes to meet a variety of injection and COVID-19 vaccine needs, each distinguished by the color of the needle hub.
Designed to meet a variety of injection needs, Lepu Medical’s Sterile Syringe For Single-Use brings improved safety to patients and doctors with its clear barrel for convenient control of foreign substances. The Sterile Syringe For Single-Use is intended only for one-time use by healthcare professionals who have received training on medical injection techniques. Injection and vaccine administrators can refer to the Lepu Medical professional manual before use for a full understanding of the device instructions, warnings and precautions.
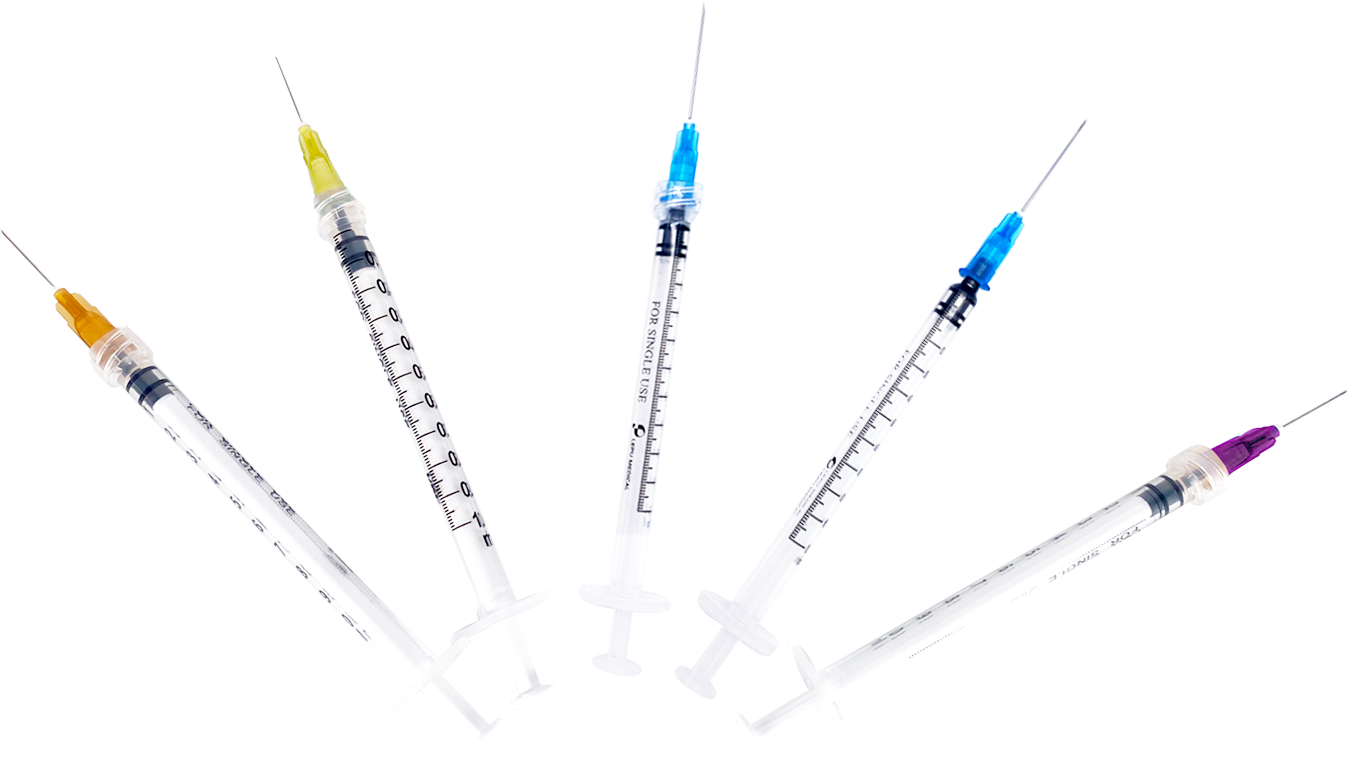
Además de la jeringa estéril para un solo uso, Lepu Medical ha proporcionado varios suministros médicos para la prevención y el control de COVID-19, incluido el kit de prueba rápida de antígeno SARS-CoV-2 para autoevaluación y el kit de prueba de anticuerpos SARS-CoV-2 para pruebas profesionales de atención médica. Lepu Medical también ofrece soluciones de diagnóstico COVID-19, como productos de ácido nucleico y soluciones para proporcionar pruebas estándar para diagnosticar COVID-19, y productos de prueba de anticuerpos para controlar los niveles de inmunidad COVID-19. Los otros productos y soluciones COVID-19 de Lepu Medical incluyen kits de recolección de muestras, kits de extracción de muestras manuales, extractores de muestras automatizados y sistemas de PCR en tiempo real para diagnósticos clínicos estándar de ácidos nucleicos.
As of August 2021, the number of global daily doses of COVID-19 vaccines continues to rise, driving demand for vaccines and vaccination supplies. According to published statistics, by the end of 2020, major governments had placed purchases for over nine billion vaccine units. With the onset of the Delta virus, vaccination rates are ramping up and the expected number of vaccine units is increasing. Government officials and medical professionals across the globe from Israel, France, Germany, the U.S, the United Arab Emirates, Bahrain, the United Kingdom, Switzerland, Cambodia and others are proposing and actively preparing for the third dose of the COVID-19 vaccination to strengthen pandemic prevention and control efforts. With the CE Mark approval for the Sterile Syringe For Single-Use, Lepu Medical will continue to proactively seek qualified partners to support the availability of reliable vaccine supplies across the globe, and assist with COVID-19 management and continuation of general medical practices.
